The Centre has published it's annual report for 2023. Read about MedTechLabs research, results and impact on society, by downloading the report as pdf.
MedTechLabs is an interdisciplinary centre for medical technology research that carries on this proud legacy to create better conditions for patient survival and improved quality of life for those with cancer, cardiovascular, and other non-communicable diseases. We create the conditions for healthcare to offer patients much more secure diagnoses and better treatment, at a lower cost.
Read the Annual Report directly on this page or download as pdf.
MedTechLabs announces a call for funding of research programmes in the areas of (1) Precision health and (2) Data-driven healthcare research.The research should be focused on medical technologies with clear clinical applications designed to prevent and treat common diseases or deal with medical needs.
Direct access to the application form: https://www.researchweb.org/is/stockholm/document/260130
Precision health aims to use data about the individual’s biology, lifestyle and environment to prevent, diagnose and treat disease with precision. Recent advancements in technology and knowledge mean that medicine now faces new opportunities to quickly obtain large amounts of biological data from patients. Using this information effectively will allow us to achieve molecularly precise diagnoses and thereby tailor individual treatments—i.e. precision medicine. Today, society’s resources are mainly spent on diagnosis and treatment, and very little on prevention. Precision medicine will provide great,cost-effective, and equal opportunities to give more people a long and healthy life.
Data-driven healthcare research: The “data-driven” concept springs from the modern technological advances that continue to produce mountains of deep, systematic, and comprehensive data. Researchers that can effectively mine the available data can discover unexpected and unpredictable relationships and develop new clinically useful knowledge. Opportunities for successful and innovative data mining comes from dramatic improvements in computing power, machine learning, AI, and other technologies.
The research programme should clearly describe the health challenge that will be addressed, the approach, how patients will benefit from the research, how the clinical studies and/or trials are expected to be executed, and the respective contribution of the collaborating KI and KTH researchers.
Available funding and deadline
MedTechLabs intends to support one or two programmes in the range of SEK 2–7 million per year and programme, during four to five years. The call opened on April 4th and closes June 11th , 2024. The estimated project start is January 1st, 2025.
Conditions for applying
- The research should be focused on medical technology with clear applications for preventing and treating widespread disease or to deal with medical needs . Planned clinical studies an/or trials shall be included.
- The programme must include one director from KI and one from KTH. Both directors must have a level of activity in the project of no less than 20% of a full-time equivalent and must be employed at least 20% by the respective administrating organisations throughout the grant period. Both programme directors must have at least a docent or Associate Professor title.
- The application should clearly describe how the programme will support careers for younger researchers.
- Patient benefit must be clearly stated.
- Patient participation in the design of studies must be included
- New applicants and those who previously obtained funding from MedTechLabs can apply. However, programme directors of on-going and completed funded MedTechLabs research programmes cannot apply.
How to apply
Applications will be processed through Region Stockholm’s portal for research applications.
Funding application instructions, opened on April 4th 2024
- Go to our website at https://www.researchweb.org/is/stockholm: https://www.researchweb.org/is/stockholm/document/260130
- Select Apply here (Ansök här).
- Fill in the parts of the registration form (registreringsformuläret).
- Fill in the application form.
Register your application on the registration form
The registration form in the system contains fields for registering details about yourself and information. It is only possible to register persons with a Swedish personal ID number or coordination number. Registered “domicile”: Enter details of the project’s “domicile”, i.e. the clinic or equivalent that will administer the project, and the hospital and department or equivalent to which the clinic belongs. The new Karolinska University Hospital organisation will be inserted into the domicile drop-down menu.
Stratipath has made the list of Sweden's best technology start-ups for several years. In just four years, their research-based AI product has been adopted by Swedish healthcare regions. It all started when MedTechLabs researchers Mattias Rantalainen and Johan Hartman were paired with entrepreneur Fredrik Wetterhall by KI Innovation.
Hi Fredrik! For those who are not familiar with Stratipath, who are you?
– Stratipath develops AI-based precision diagnostics that help doctors in the healthcare sector to choose which treatment a cancer patient should receive. There are many challenges in healthcare, we work in the field of cancer and there are many good screening programmes, for example for breast cancer. But many patients end up in categories where it is not possible to assess how aggressive the cancer is. The analyses that can answer this question are expensive and time-consuming, so not all patients have access to them. This leads to inequalities in healthcare, as your treatment and diagnosis may vary depending on where you live or which healthcare provider you see. So, in situations where it’s clear that you have cancer, but you can’t judge how aggressive it is, that’s where we come in with a diagnostic solution based on advanced artificial intelligence and deep learning. Our method is simple, cost-effective, and fast. Compared to current methods, which can take up to several weeks, our analysis takes less than 30 minutes. Right now, we have a customer who is starting up with our solution and their current analysis takes an average of 20 days. In the meantime, as a patient, you are waiting for your judgement. This time is extremely stressful for patients. And for the relatives, it can be added. It also affects the work of the health service, which is keen to get treatment started. Having to reschedule patients and pick up the case again, where it is necessary to read up on the patient’s illness once more, creates a lot of extra work.
Do the people you meet understand what you do?
– When we meet our healthcare customers, I find that they immediately understand what we do and the value of our solutions. Others may find it a little more difficult to understand as the diagnostic and oncological workflow is complex and not something you see unless you work in it. What many people encounter and perhaps most commonly associate cancer diagnostics with is screening/mammography, which aims to detect suspected cancer. It is the first and only one of many steps before diagnosis and treatment. We come in when the cancer is detected, and the basic diagnostics are done and the important decision of what treatment to give the patient is made.

Fredrik Wetterhall, CEO and co-founder of Stratipath.
Where do you stand now and what has brought you there?
– We are currently in the situation where three Swedish regions are using our solution in their diagnostics, and we will most likely get more healthcare providers during the spring. Then we are looking at starting up sales in other parts of Europe, mainly northern Europe. We started in 2019 and we entered clinical operations in April last year, so we have been around for four and a half years. There were three of us who started the company, and the first employee came in 2020. Now we are 16 people working here at Nanna Svartz väg in Solna. During these years, we have learnt more about our product, made updates, validation studies and started to develop more applications outside the breast cancer area. The big challenge we face now is to roll out internationally.
It sounds like things have gone pretty quickly, for being a life science company?
– It is always necessary to have a regulatory approved solution, it was and is a large and extensive work to be able to quality assure a product. We were fortunate that when we started the company, a lot was already in place. I, Johan Hartman, pathologist at KS and professor at KI, and Mattias Rantalainen, associate professor at KI, met through KI innovation. Johan and Mattias and their research groups had been collaborating for many years already, so at that time they already had promising results, so we didn’t start with a blank white paper and that’s why we were able to get a product out relatively quickly.
Where will Stratipath be in five years?
– We will be a well-established company and present in the main markets, in Europe and North America. But we are already receiving enquiries outside these regions, so we will certainly have established ourselves in other markets too. In five years, we will have a broader product portfolio, both in breast, but also in other major solid cancer diagnoses such as prostate, colorectal and others. We focus on the major cancer diagnoses as our technology requires access to large amounts of data, whereas our technology is not as well suited for more rare cancers. At that point, we offer both prognostic but also treatment-predictive solutions. An important part of our roll-out is collaborations with larger companies, such as Sectra and the US-based Prosia and Paige. There are also other corporate partnerships that are in the pipeline but which I cannot mention now. This is an important part of our ability to have a global impact; these partnerships allow us to reach more patients. In five years, we will be over 100 people and need to increase the team both in R&D and the commercial side. Today, most of the cost base is in R&D. We will not fit in these premises either. Our ambition is to remain in Hagastaden, but we will probably also have sites outside Sweden. So that we are close to our customers and to reach expertise in other parts of the world. Because it is not possible to recruit only here in Sweden if we are to have the sharpest expertise.
What does Stratipath mean in a larger perspective for Swedish medtech?
– I can probably start with the healthcare sector. We can contribute to more equal and sustainable healthcare. Because it is unequal today. There are different treatments and there are many under development, many of which are good ones. But not all treatments can be given to all patients, there are costs and side effects that prevent it. We can contribute to a more cost-effective healthcare system, and we can help healthcare providers to give their patients the right treatments faster. Hopefully, our journey can create interest for other researchers and clinicians to take their research all the way to the patient, inspiring them to commercialise their results. Also, there is no doubt that we have built a great competence here in Stratipath’s team that I believe will benefit other medtech companies.

Delar av Stratipaths teamet på kontoret i Hagastaden.
What is needed for you and similar companies to develop faster than today?
– Access to capital – the investment climate in the US and in Europe is so different from here. Companies there raise five to ten times more capital than we can in Sweden, which means they can develop faster. This has become an even greater challenge because of the wars and unrest in the world, which has reduced the risk appetite of investors even more, and many investors are also afraid of the healthcare sector because they know that it is difficult and takes time to introduce new technology. Then we need expertise. In our field, we see that there is good expertise in Sweden, but there are other countries that are investing even more. So, we are competing for talent with Asia, Europe and the USA. If we as a country want to continue to be a world leader in our sector, for example, we need to ensure that we have world-leading research and education, and that it is attractive for talents to stay in, or move to, Sweden. One thing that we in the business community need to take responsibility for is that we really understand healthcare and the problems and challenges that exist and really need to be solved. Here I think there is potential for improvement, and I think the key is that companies should be started around the problem they want to solve and not around a technology they want to find a use for. I also believe that Sweden must ensure that the healthcare sector has better opportunities to embrace new technology. But this is tricky because their focus should be on taking care of the patients, and I think many in the healthcare sector are tired of various IT projects that never make it past the pilot stage. For us, it is important that our solutions are tested and evaluated in a real environment and not in some side business, only then can we get real feedback. Here we have been successful, but I know many who have not. The next challenge is to scale up use, which usually requires changes to guidelines and reimbursement systems. Changes to these are often quite long processes that take many years, requiring resources and a stamina that not many smaller companies have. This means that many healthcare providers are stuck with old technology when there are both better and cheaper alternatives. This is not good for healthcare, patients, nor companies. Here it would be desirable to be able to make faster changes, which would benefit everyone. Of course, we should not compromise patient safety or set requirements for clinical evidence, but I believe that everyone in society would benefit from faster introduction of new technology in healthcare. In Germany, DiGA has been introduced to speed up the introduction of certain types of new digital solutions. This is far from solving all problems, but perhaps it could be something for Sweden to follow?
What is your background and what made you decide to invest in building Stratipath?
– I once started at Siemens Elema, developing intensive care ventilators. What made me stay in the healthcare sector was that I need a clear and good purpose for what I do. I’ve had a hard time finding that in any field other than the health sector. I started with software development in the 90s and for the past 25 years I have been involved in commercialisation and implementation in healthcare, both on the supplier and provider side. I find it very stimulating to develop new products in healthcare and see how it creates radical improvements for both individual patients and the healthcare system. During my career, I have started several companies based on research from KI. It was Mats Ferm at KI innovation who matched me with Johan and Mattias, and together we started thinking about how we could find a model so that the results of their research can create value for healthcare and patients. At first, we thought that this could be done through a larger established company, but we realised that we could achieve greater impact by starting our own. We took a liking to each other, where we saw that we had a good competence together, commercially, clinically, and technically, and that’s the way it went. I am impressed by Johan and Mattias as clinicians and researchers who dare to invest in entrepreneurship in this way. Because this is a new world for them, to take the step from research and healthcare and put time and energy into building a company. Because it should be recognised that it is not always easy. Their driving force is that their research should benefit patients.
What are you most looking forward to in 2024?
– Getting out more widely and reaching more patients with our solutions. Learning more about how our product is used in healthcare, in Sweden and internationally. Seeing the impact that the product can make. That’s the most rewarding thing for you, to see it being used and making a difference. There are so many companies, projects and products that fail. It is also very stimulating to work in this area, with the rapid development that is taking place. When it comes to the team, it’s an honour to spend time with driven, highly competent people. I am particularly proud that we have an even gender balance, many nationalities, ages, and backgrounds. I think everyone who works with us is doing it for a higher purpose. It simplifies recruitment, and it makes it easier to get out of bed with a smile and go to the office even when it’s dark and snowing outside. Passion is central and important for everyone to be able to cope. We are not running a short race, we know that we will have to fight for many years.
Facts about the company:
Stratipath is a Swedish company that develops AI-based precision diagnostics with the aim of radically improving cancer diagnostics and the conditions for everyone affected by cancer. Stratipath’s AI solutions analyse tissue samples from tumours and enable more precise treatment decisions through improved and faster patient stratification in healthcare, clinical trials, and drug development. This means that more patients can have access to precision diagnostics, which contributes to more equal and sustainable cancer care. Stratipath was founded in 2019 and is based on world-leading academic research at Karolinska Institutet. www.stratipath.com
In Sweden, more than 18,000 people suffer an acute ischaemic stroke every year. With correct and rapid treatment of stroke, both lives are saved, and the affected person can avoid lifelong disability, something that both provides increased quality of life for the patient and saves big money for society. The new treatment options that have emerged over the past decade require more knowledge in healthcare, which is one reason for the new course offered through MedTechLabs.
The course “Acute stroke, diagnostics and treatment” organised by MedTechLabs started in October 2023. The course is aimed at residents and specialists who want to train in this area to better follow the national recommendations and guidelines for examination and treatment of stroke and TIA. The new course was offered to doctors in Region Stockholm as well as in other parts of the country and was quickly oversubscribed.
We spoke with Håkan Almqvist, senior physician and neuroradiologist at Capio St Göran, who is responsible for the course. Håkan has worked almost all his professional life with stroke, first at SÖS and later as a neuroradiologist at Karolinska and has followed the modern development of how to find and eliminate thrombi/embolisms since the late 90s.
Hi Håkan, can you tell us a bit about the course and why it is given?
“The course aims to provide knowledge about the benefits of acute reperfusion therapy within 24 hours in several patient groups, including those with relatively large established infarcts. The management and assessment of TIA is also included in the course. “The biggest problem with stroke care is still that patients come in too late, so it’s important that everyone in the community knows that help is available and picks up the phone if they have symptoms. But we in the healthcare chain also need to be on our toes and know what is needed for the patients. We must dare to be rational and inclusive, even if there is a low probability of stroke. Because it can be difficult to know whether your judgement is correct. Doctors need to see many cases to become skilled in their judgement and that takes time, especially in a smaller hospital.”
How do you deliver the course?
“In the first round, we offer it free of charge to 40 participants who receive around 30 video lectures and case study material with commentary, which we distribute using Collective Minds Radiology and their radiological imaging system. It is up to the student how much time they spend on that part, but I would say it is about two and a half days, at their own pace. In addition to this, we have a mandatory physical meeting in the form of a seminar in Stockholm where case studies are discussed. The knowledge check takes place at this seminar and active participation is required to obtain the course certificate.”
Who came up with the idea for this course?
“Many of us realised early on that there was a lack of continuing education for the new forms of examination and treatment that have emerged in the field. When MedTechLabs started, it was realised that there was a need for training in the region. Professor Staffan Holmin, together with KTH professor Mats Danielsson, saw an opportunity for the centre to contribute to and provide training. “As early as 2018, we started recording parts of the material that is now used in the course, along with new material in 2023. The pandemic and questions about which platform to use have meant that it has taken some time.”
What do you think has made the course so popular?
“The course only corresponds to a few days where you choose when for about 1 month. That certainly has an impact. But it is up to the student how much time they spend. I think long courses are becoming more difficult to attract. That we have a hybrid approach with self-study with lectures and being able to browse cases combined with a physical seminar with the teachers. A course must also be able to provide both a course certificate and an ST certificate. This is probably exactly what many doctors are looking for in terms of continuing education.”
When will you hold the course next time?
“This is the first time we have held it, and we will of course further develop it based on the feedback we receive from the course participants. Then we will see how many times a year is reasonable. It is also a question for Region Stockholm, which this time allowed doctors from other regions to attend free of charge. But hopefully we will also organise the course in 2024.”
MedTechLabs has granted SEK 35 million for two new medical technology research programmes. The first will develop a new method for monitoring fetal oxygenation during labour. The second programme will develop a new method for early diagnosis of brain diseases using MR elastography.
This year’s call from MedTechLabs was launched in the spring and the decision to adopt two new research programmes was taken by the centre’s steering committee in early October. Johan Schuber, Executive Director of MedTechLabs, says that the approved programmes strengthen the centre’s focus on conducting clinically relevant research.
Current fetal monitoring methods have weaknesses and the researchers in this new programme aim to improve them to reduce the risk of organ failure in the baby during delivery. This is an important area of research that affects many patients and maternity services, not only in Sweden but also worldwide.
The programme ‘Continuous lactate measurement and prevention of fetal hypoxia during delivery’ is led by Malin Holzmann, Karolinska Institutet (KI), and Saul Rodriguez Duenas, Royal Institute of Technology (KTH). Malin Holzmmann is a senior physician at the Unit for Pregnancy and Childbirth at Karolinska University Hospital in Solna, Sweden, and an associate professor at KI. Saul Rodriguez Duenas is a senior lecturer at the Division of Electronics and Embedded Systems at KTH. The goal is to achieve continuous lactate measurement during labour, which would represent a completely new technology in healthcare with the potential to fundamentally change fetal monitoring.
The second research programme focuses on understanding how brain tissue changes when exposed to diseases such as Alzheimer’s, Parkinson’s, MS, or brain tumours. With the increasing availability of drugs to treat these diseases, early detection of the diseases in time is becoming more important to slow down disease progression as effectively as possible.
The programme “Clinical application of next-generation brain magnetic resonance elastography for neurodegenerative diseases and brain tumours” is led by Rodrigo Moreno, KTH and Tobias Granberg, KI. Rodrigo Moreno is Associate Professor and Senior Lecturer at the Department of Biomedical Engineering and Health Systems at KTH. Tobias Granberg is Senior Lecturer at the Division of Clinical Neuroscience at Karolinska Institutet. MR-Elastography (MRE) is already used today to diagnose liver diseases, but its complexity has so far hindered its clinical use for the diagnosis of brain diseases. The main objective of this multidisciplinary programme is to enable MRE to be performed also for the brain and to evaluate its clinical utility in neurodegenerative diseases and brain tumours. The project covers MRE technology from image acquisition and analysis to its use and evaluation in clinical trials. The goal is to enable clinical implementation in Region Stockholm within five years, thus contributing to better treatment for patients.
Research in both programmes is expected to start in January 2024.
MedTechLabs is an interdisciplinary centre for patient-oriented research that will contribute to breakthroughs in the development of medical technology with significance for the important challenges of healthcare and our major public diseases. In all programmes, researchers and clinicians with both technical and medical expertise collaborate. The centre is run jointly by KTH, Karolinska Institutet and Region Stockholm.
For more information, contact:
Johan Schuber
Executive Director of MedTechLabs
Phone: 08-790 67 64
E-mail: jschuber@kth.se
Website: www.medtechlabs.se
LinkedIn: https://www.linkedin.com/company/medtechlabsstockholm
SVT – the Swedish national broadcasting company – highlights the impact of research on AI for breast cancer detection in mammography, conducted by MedTechlabs researchers Kevin Smith and Fredrik Strand. Also, Lancet published a scientific article on the study conducted at Capio Sankt Göran Hospital in Stockholm.
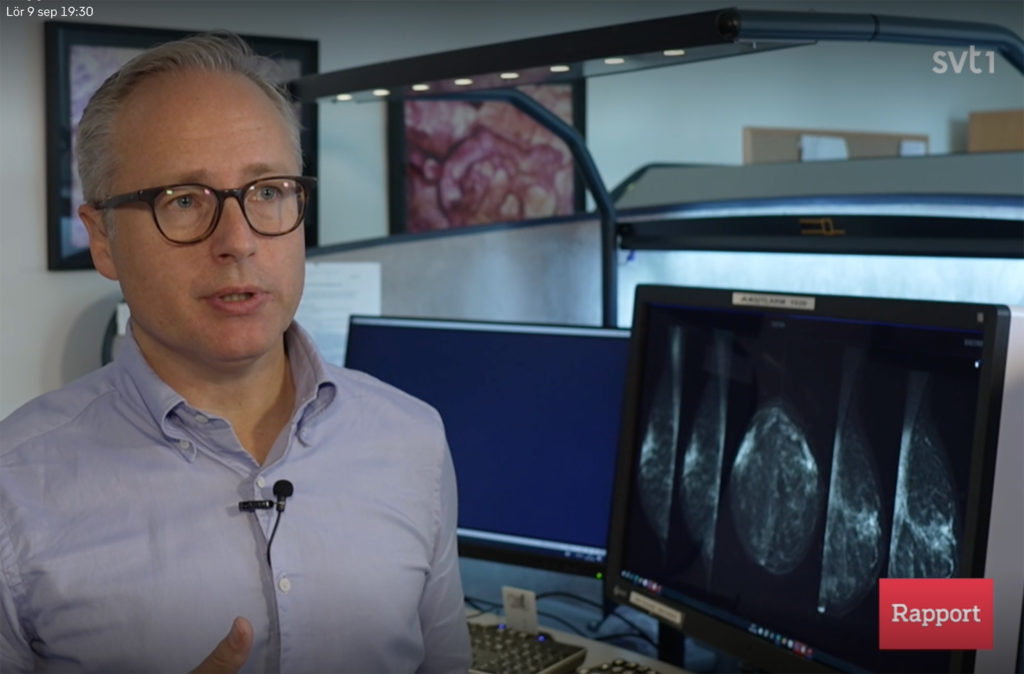
What differences in cancer detection and unnecessary recalls are the results of various combinations of AI and radiologists? Lancet Digital Health has published the results of a prospective clinical trial conducted in Stockholm.
MedTechLabs researcher Fredrik Strand is surprised at how accurate the AI was and says that the potential for AI to handle most of the screen-reading is huge.
Karin Dembrower is chief physician and medical director of the Mammography Department at Capio S:t Göran’s Hospital where the clinical study was conducted. She is very satisfied with the results.
– We found slightly more cancer and recalled fewer women who turned out to be healthy, so we made fewer women worried. And we freed up time for the radiologists to do other diagnostics than looking at mostly healthy women’s images. We have managed to cut the queues considerably so we are a queue-free breast centre now, she says in the feature.
The AI-assisted breast cancer detection system has been operational since summer 2023 at St Göran’s Hospital in Stockholm.
– We have shown that our AI technology works on St Göran and their equipment. But that doesn’t mean that any AI will work on all equipment, so it’s something you need to check before introducing it, says Fredrik Strand in the same feature.
Watch the tv-feature here (in Swedish)
Read the Article in Lancet Digital Health here (full text)
On the 24th of August MedTechLabs were proud to be among the medical research facilities in Stockholm and Uppsala to host a visit by GE Healthcare Top Management. Their team was happy to see the progress of the world’s first clinical evaluation of the Deep Silicon CT detector technology for photon counting CT here at BioClinicum in Hagastaden.
Peter J Arduini, President and CEO at GE Healthcare later stated:
“I spent time in my early GE HealthCare career working in CT. This week, it was thrilling to be at the epicenter of this exciting innovation, and I look forward to the potential transformation it will create for CT and the benefit for patients worldwide. Photon counting has the promise to further improve the capabilities of traditional CT and increase imaging performance for oncology, cardiology, neurology, and many other clinical CT applications.”

On 19 April 2023, MedTechLabs organised a seminar focusing on strategies for researchers to better identify and protect their digital innovations. Also on the agenda were tactics and practical tips on patenting or other ways to extract business opportunities from science linked to digital applications.
Niclas Roxhed, director of MedTechLabs, began by giving a background to MedTechLabs and its focus on interdisciplinary, translative research in later phases, proximity to clinical trials and clinical implementation, and industrial collaboration. He then gave the floor to Måns Marklund, IP strategist and founder of Cascelotte.
Måns then explained how he and his company have analysed results from researchers linked to MedTechLabs, examined the volume of patents and also made an in-depth study of their quality, potential economic value and technical standard. Compared to some of the world’s best research centres, these patents were found to be of a very high standard, especially those linked to Mats Danielsson and his research teams.
– Other research at MedTechLabs turned out to be more linked to digital applications and artificial intelligence, which may be one reason why this kind of research does not lead to as many patents. After discussions with Johan Schuber, Executive Director of MedTechLabs, we therefore decided to investigate this phenomenon more closely, said Mats.
How good is Sweden in international comparison?
Mats explained the importance of looking at the differences between different kinds of technologies. Normally, Sweden ranks 12th in the world in terms of all technology patents. However, many come from Ericsson, which inflates the volume. In life science alone, Sweden is ranked 15th, which is still very high, and an indication of how good Sweden is even without Ericsson.


– In AI, Sweden is ranked 20th in terms of patents, also very high considering we are a small country. But perhaps there is room for more patents, with increased knowledge of how this works and why patents should be granted in this area as well. We also know that if there are patents in an area, there are many other intangible assets there. It could be trademarks, knowledge of data and data use, design and so on. So, the patents indicate a country’s ability to transform innovation into economic value, Mats explained.
He went on to add that AI is also one of the most patented assets now, so it is not true that AI cannot be patented, even if it is slightly different from other technologies. This also applies to the life science area in general, compared to other patent areas.
– It is important to understand which strategies, tactics and practical approaches are good to know when it comes to both AI and life science. Therefore, we chose to invite Michael Kitzler and Malin Keijser Bergöö, who both work as lawyers specialised in European patent issues at Rouse, said Mats and handed over to these speakers.
Positions of control and the commercialisation of innovation
– In a commercial context there are some fundamental questions you need to ask yourself, like what you’re selling – sometimes it seems like you’re offering one thing when it’s actually something else. In the context we are talking about now, many of you are probably in an early phase of commercialisation, maybe you have created a company and you have something to sell to someone, Michael began.
He then described how Rouse usually starts by identifying the intellectual assets that the research team or company has built up over time and then translates these into different “control positions”, which he explained as the aspect of control that creates ownership and/or control over an innovation over time. Some of these can be patented, some may need to be kept secret until later, some can be contracted with other actors, and some are based on a unique combination of capabilities that are difficult for others to acquire.
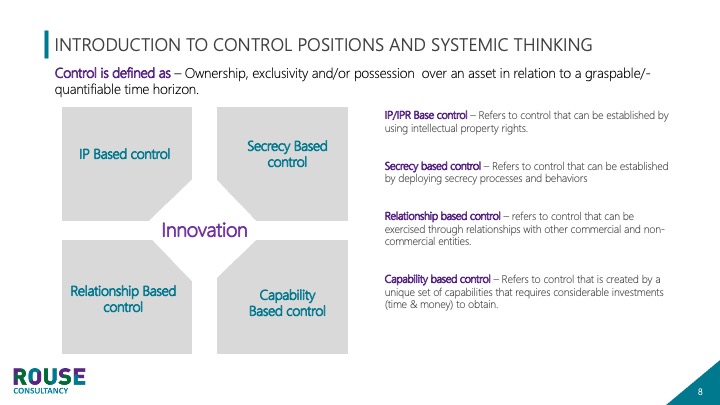
He explained that different companies/teams need different mixes of control over assets and gave the example of a company that had 55 per cent of its assets covered by patents, 25 per cent by external agreements of some kind and 10 per cent by secrecy-based control. Software and IT companies tend to have fewer patents and are more likely to use other methods, while high-tech companies put a lot of effort into getting patents. Michael argues that medtech companies are somewhere between high-tech companies and software companies in terms of control positions.
– Looking at the trends in medtech, the statistics show that there are more patent applications in this industry than in others. So, is it worth the money to make these applications? Probably, said Michael.
The largest patent owners in medtech include companies like Siemens Healthcare, Philips, Fujufilm, Canon Medical Systems and Merative. Then there is a “long tail” of companies that may not have as many patents, but these can be very important. Michael gave the example of Elekta, with only 12 patents in the field of AI, but which are very significant. Even further down the list, there are companies with maybe four patents, which can be very relevant portfolios despite their small number. To summarise, it doesn’t take many patents to make a difference and achieve commercial success, it’s all about the content of those patents, Michael explained.
Michael then listed the commercial values that patent control positions create for medtech companies, from limiting business risks to contributing to the company’s participation in key ecosystems (see image below).

Patenting medtech and AI in practice
– Your field is one of the toughest to patent. Partly because it has to do with things you do with the help of computers, which the patent authorities are very careful about before approving, which makes it complicated and requires more work and the right strategies, Malin began.
She explained that in some countries there is a ban on patenting treatments, which can affect companies, and that you are then allowed to patent the devices you use, not the methods. This differs depending on where you are in the world; in China, for example, it is easier to patent treatments. In Europe, on the other hand, it is often impossible to patent treatment methods. In the US it is more unpredictable, where a single court decision can change the situation so that it suddenly becomes possible to patent something that was previously impossible, and vice versa. Malin emphasised that having said that, it is still often possible to patent – in some way. You may have to go through the equipment instead of the method, or do it in other ways, but it is always possible to patent something.
– One thing we saw when we looked at medtechlabs was projects in bioelectronic medicine where inflammation is reduced by electrical stimulation via an implant. If you want to patent at an early stage here, the concept constitutes a treatment method, which means that you might choose to patent the implant instead. But competing companies may then choose to use a different implant. So very broad concepts can be difficult to patent with commercial success, Malin explained.
She then gave an example of pacemakers. A pacemaker stimulates the heart and is therefore considered a treatment, which cannot be patented in Europe. But the actual method of identifying which signals the pacemaker should use to stimulate the individual patient’s heart in the right way can be patented – if you state that you store this data somewhere and do not use it directly to stimulate the heart. Then, of course, you can also use this stored data to stimulate the heart. An example of how you can adapt the design of patents to the legislation and get an effective patent that provides indirect protection for the treatment method anyway.
Malin also gave the example of a company that has patented a detailed description of how they trained their medical image recognition algorithm, thereby gaining intellectual property protection for their innovation. This protection has been granted worldwide, including in Europe. Another example involved wristbands used to alert an elderly person to a fall. The company used AI and machine learning to correctly interpret the signal from the bracelet. The solution to obtain patent protection here was to focus on specific parameters that were used when the algorithm analysed the signal to distinguish between an accident and the wearer just lying down. Such parameters could be, for example, which room the wearer was in, whether the wearer had moved, slept recently, run, etc. The combination of parameters provided a basis for the way the algorithm works, and this was the basis of the patent application.
– One challenge when you want to patent AI solutions is that you can’t always describe why the solution gives the answers it does, because they are based on an algorithm that has trained itself to find the answers on huge amounts of data, but in patent applications you have to be able to describe this, said Malin.
Michael summarised the need to contextualise AI and gave some key takeaways: patenting can be a powerful tool to control the value-creating phenomena that arise in digital medical technology. However, a systematic approach is required by those who want to create control through patents and other control measures. Finally, patenting in digital medtech is more complex than in most other fields and therefore requires well thought-out strategies. To achieve powerful patents in medtech, you must do the right thing, which often requires a more systemic approach. You also need to understand the full range of assets you have and how they can be used and controlled, concluded Michael.
Questions from the audience
Niclas then moderated questions from the audience, to which the participants provided answers.
Question: What is required to be able to patent an AI model for radiological images?
Answer: The way to build and train the model, e.g. based on specific parameters, if these are very different from other models, then it is easier to patent it, provided it is new. Just using a general model trained on many images is more difficult.
Question: What happens if a researcher has published an academic paper describing such a method?
Answer: It depends on how long ago it was published. In the US, you have one year to apply for a patent after publication without blocking yourself from patenting. In Europe, you must apply before publication. It should be added that academic papers do not always disclose the whole innovation, they can be more general and do not necessarily describe details that make patenting impossible, in Europe.
Q: What happens if you have patented and published something and then want to patent something that is slightly different from what was first patented?
Answer: It always gets complicated if you change things in a follow-up patent application. It is better to have everything worked out and right from the start. But the reality and needs of publishing an innovation may take priority and you should generally file an early patent application; it is better than not claiming your innovation at all.
Q: What does protection mean in practice if a large company infringes?
Answer: Unfortunately, those with the most money win. But if you have a patent, you can enlist the help of another big company to help you win against the infringer. The patent can also act as a deterrent to large companies who don’t want to get into trouble. Large companies may also appreciate that a smaller player talking to them has a patent for their innovation, so that it is clear what it is about.
Question: You can’t patent data, but can you patent the way you curate data when training an AI?
Answer: It depends on whether the approach is very different from how the same thing has been done before. If it is done in a different way, it can work, compare with the example of the bracelet that would warn of falls, where the choice of parameters was part of the curation. Then there is the actual database protection, that you own this data. And when you process the data, a new data set is created, much like the translation of a literary work from Swedish to English. In some cases, it can be difficult to prove who originally owned the data. The EU also requires that certain health data must be made available to other users, known as data sharing.
Q: Does a patent application need to have proof that the innovation works?
Answer: No, normally not. A classic example is the patent protection of perpetual motion machines.
Question: Can recommendations for treatment be patented?
Answer: Yes, the example of the pacemaker is such a case, where the signal to be used for the treatment was first stored and then used.
Question: Is it common for sufficient preparatory work to be done on IP at the pre-commercial stage? Answer: Yes, I would say so (Michael). But you should always make a business case before applying for a patent, because it can cost around one million to do all the IP work, and then you need to know that you will earn at least that amount to make it worthwhile (Malin).
KTH Innovation: we can help in these phases too, for the researchers who need it.
Nicklas: And KI Innovation can also contribute in a similar way.
Niclas: and to present a credible business case, you need to try in the IP area beforehand. This is at least my experience from this area.
Michael: And we’ve said here today that all patents can ultimately be broken by big players, but if you don’t have a patent right from the start, you have no control over what you’ve come up with and no negotiating position.
Niclas: What belongs to the field of medtech is also the long lead times, not least when the innovation is to be tested clinically and then implemented, generating the need for a lot of additions to the patent application.
Question: What are the three most important things to consider for a research group that has developed an innovation and wants to commercialise it in the future?
Niclas: To patent at all. I always suggest it to my postdocs – for every paper, the legal requirement that it is an innovation is there. There should be potential there. For every paper we publish, we submit a patent application, in principle. You also have to consider whether you can get your money back on such an investment. If you don’t see that possibility within a year, you might drop the application.
Question: We talked earlier about how you can’t protect a biomarker, but you can protect technical equipment. Nor can you patent a single molecule, but is it possible to patent a combination of molecules?
Malin: It depends on the country. In the US, you used to be able to patent treatments using a combination of biomarkers, but that is no longer possible. Nowadays this is considered a natural phenomenon that cannot be protected.
Question: how to protect AI innovation in early collaborations between researchers and companies. How do you protect the agreements you make there, when you may not even have published anything?
Michael: This is an important phase where you need to define the origin of the innovation through agreements. Even if it is too early to apply for the patent, the agreement is valid in court. Unfortunately, research teams often settle for agreements that are too general and do not go into the details of who owns what and how this will be handled at a later stage. Again, researchers need to get proper help from lawyers who know this area, because creating these agreements is complicated. I have seen so many cases where it has gone wrong.
Question: How broad can patents for diagnosis based on AI be?
Answer: It depends on whether you put in things like what parameters you used. Just using general descriptions that you used AI is not enough. Then your patent may only cover, say, 17% of the solution you developed. But then you still have protection for that part of the window. Many patents are quite narrow, but if you have the smartest solution, the market will probably not settle for an inferior solution just because it is not protected. And then you have something to sell.
Do we have the right organisation for highly specialised care in Sweden, with seven university hospitals competing for highly specialised assignments?
That question was debated at the Almedalen seminar where Staffan Holmin and Mats Danielsson participated: “From frontline to routine care. How to create the cutting-edge healthcare of the future?”. The panel also included Acko Ankarberg Johansson, Minister of Health, Björn Zoëga, Hospital Director, Karolinska University Hospital, and Annika Östman Wernerson, President of Karolinska Institutet, Olivia Wigzell, Director General, National Board of Health and Welfare.

The seminar was organised by the Karolinska University Hospital and Dagens Medicin Sverige. Nina Hedlund was the moderator. Here is a web link to the seminar (which was held in Swedish): https://lnkd.in/gih5QWay
In episode no 121 of Karolinska Institutet’s podcast Medicinvetarna, Magnus Boman gives his views on potential applications and ethical challenges in the use of AI in medicine and research.
On behalf of the MedTechLabs steering committee, Magnus Boman is mapping healthcare needs in terms of AI, and possible synergies between research centres and suggests training courses in the area that MedTechLabs should be responsible for.
You can find the episode here (in Swedish).


